Heidelberg Pharma AG
Wird der Staat Unternehmer? Das ist noch nie gut gegangen. Ich denke, man sollte die F&E Kosten als Zuschuss oder Darlehen vergeben. Die EIB ist ja auch noch da. Vermutlich hat die USA da im Hintergrund Druck gemacht. Prompt kommt ja schon die Antwort aus USA mit dem Abzug von 12500 GI´s. Die Aktion halte ich nicht für gut. Wenn Herr Hopp doch da die Mehrheit hat, müßte er in der Lage sein, einen Verkauf zu verhindern. Irgendwie läuft da im Hintergrund etwas. Warum beteiligt sich dann der Bund nicht an weiteren Perlen wie Morphosys, HP, Evotec etc? Eventl. werden anstehende Zulassungen (z.B. Remimazolam Cosmo/Paion) in USA verzögert oder verhindert. Das Ganze ist nicht nach meinem Geschmack.
Zudem, wer sagt schon genau, ob das Verfahren bei Curevac klappt? Es sind ja einige Unternehmen dabei. Da ist eine Investition in einen Biotechindex vielleicht die besser Wahl.
War mal in HP investiert (wegen der anderen Wirkstoffe). Mit gutem Gewinn vor kurzem raus und überlege momentan einen Einstieg. Das heute hat mich allerdings zurückgeworfen. Ist mir zuviel Hype.
Ich weiß gar nicht wieso das Thema Curavec hier schon wieder so wichtig genommen wird. Hat mit HDP doch nix zu tun. Auch beim letzten Mal hatten wir die Diskussion schon. Da kam der Kursanstieg durch Redhill und nicht durch Curavec.
Ich bin mal gespannt, ob ich Recht habe.
Curevac erhält Genehmigung für klinische Corona-Impftoffstudie in Deutschland.Quelle: Guidants News https://news.guidants.com
Melbourne (Australia) – 18th June 2020. Telix Pharmaceuticals Limited (ASX: TLX, ‘Telix’, the ‘Company’)
today announces that the Company’s ZIRCON phase III trial of TLX250-CDx (89Zr-girentuximab)1 has
recommenced patient recruitment in Europe, following a pause to clinical trial activity as a result of the
COVID-19 pandemic.
2
Patient recruitment to the ZIRCON trial has recommenced at clinical sites in France with the dosing of
two patients with TLX250-CDx this week at Centre Hospitalier Universitaire (CHU) de Nantes (Nantes,
France). Clinical sites in Belgium and the Netherlands have also been reactivated, with patient
recruitment expected to resume in the next two weeks. Telix expects clinical trial sites in Australia,
Canada, Turkey and USA to follow between now and September, subject to conditions remaining stable.
Telix CEO, Dr Christian Behrenbruch stated, “We are relieved to be restarting patient recruitment into the
ZIRCON study and gratefully acknowledge the patience and dedication of our investigators and their
clinical staff during this necessary hiatus. Although we had stopped patient enrolment, we had not paused
recruitment and we see an encouraging patient backlog to regain momentum.”
About the ZIRCON Study
ZIRCON (Zirconium Imaging in Renal Cancer Oncology, NCT03849118) is an international multi-centre
Phase III study at 33 sites in Europe, Australia, Turkey, Canada and the United States (subject to
regulatory approval in the various jurisdictions). ZIRCON is a prospective imaging trial in approximately
250 renal cancer patients undergoing kidney surgery, to determine the sensitivity and specificity of
TLX250-CDx PET imaging to detect clear cell renal cell cancer (ccRCC) in comparison with histologic
“standard of truth” determined from surgical resection specimens.
https://www.dgap.de/dgap/News/corporate/...vestor-day/?newsID=1357045
Der Investorentag heute scheinbar nicht ;-)
Evtl. Morgen ;-)
Über Heidelberg Pharmas firmeneigene ATAC-Technologie
Antikörper-Wirkstoff-Konjugate (Antibody Drug Conjugates - ADCs) kombinieren die hohe Affinität und Spezifität von Antikörpern mit der Wirksamkeit von kleinen toxischen Molekülen, um Krebs zu bekämpfen. ATACs (Antibody Targeted Amanitin Conjugates) sind ADCs, deren Wirkstoff aus Amatoxin-Molekülen besteht. Amatoxine sind bizyklische Peptide, die in der Natur im Grünen Knollenblätterpilz vorkommen. Durch Bindung an die RNA-Polymerase II hemmen sie die Transkription der mRNA, einem Mechanismus, der entscheidend für das Überleben von eukaryotischen Zellen ist. In präklinischen Studien haben ATACs eine sehr hohe Wirksamkeit gezeigt, sie überwanden häufige Resistenzmechanismen und können auch ruhende Tumorzellen bekämpfen. Quelle: https://www.dgap.de/dgap/News/corporate/...vestor-day/?newsID=1357045
Danke GOLDFISCHREGEN für das Posten des Link.
Telix Granted FDA Breakthrough Therapy Designation for Renal Cancer Imaging Product.
Girentuximab (TLX250-CDx) ist von HP an Telix auslizensiert .
Melbourne (Australia) – 1st July 2020. Telix Pharmaceuticals Limited (ASX: TLX, ‘Telix’, the ‘Company’) today announces that the US Food and Drug Administration (FDA) has granted Breakthrough Therapy (BT) designation for Telix’s renal cancer imaging product TLX250-CDx ( 89Zrgirentuximab). Under BT status, the FDA will work closely with Telix to provide guidance to the Company on the development of TLX250-CDx for the diagnosis of “indeterminate renal masses” that have been identified on CT or MRI imaging.
TLX250-CDx is being developed for the purpose of determining whether such “indeterminate renal masses” are either clear cell renal cell cancer (ccRCC) or non-ccRCC, using Positron Emission Tomography (PET) imaging. BT designation offers a number of significant benefits to Telix, including eligibility for Fast Track designation, more frequent and intensive interactions with the FDA, and the opportunity to submit a “rolling” Biological Licence Application (BLA) for TLX250-CDx, where the application can be submitted in separate modules to streamline the FDA review process for approval.
The criteria for BT designation require preliminary clinical evidence that demonstrates the product may have substantial improvement on at least one clinically significant endpoint over available care.
Telix CEO Dr Christian Behrenbruch said, “The granting of Breakthrough designation by the FDA for our kidney cancer imaging product provides Telix with the opportunity to interact closely with the FDA to expedite the registration process of TLX250-CDx, a particularly important consideration given the current Phase III development status of the asset. There is a significant unmet medical need to improve diagnosis and staging of clear cell renal cell carcinoma (ccRCC), which is the most common and aggressive form of kidney cancer. It’s encouraging that the Agency recognizes this.”
Heidelberg Pharma AG | wallstreet-online.de - Vollständige Diskussion unter:
https://www.wallstreet-online.de/diskussion/...e/heidelberg-pharma-ag
https://langfristanleger.net/...analyse-der-heidelberg-pharma/aktien/
GIRENTUXIMAB ist ein Antikörper, der zusätzlich mit einem radioaktiven Teil bestückt wurde. Wilex nutzte dafür Iodine (124I). Dieser Wirkstoff sammelte sich im Krebsgewebe des Nierenkrebses an und konnte mithilfe von diagnostischen Geräten sichtbar gemacht werden. Bei der Sensitivität verpasste Wilex damals knapp den erforderlichen Wert. REDECTANE wurde im Januar 2017 an den australischen Spezialisten Telix auslizensiert. Dieser Spezialist optimiert nun den Antikörper mit einem neuen radioaktiven Teil 89 Zirconium zu 89Zr-girentuximab.
Eine kleinere Bridging Studie zur Bestätigung der Verbesserung wurde von Telix durchgeführt. Nachdem diese Studie eben diese erhofften Merkmale eindrucksvoll unter Beweis stellte, startet nun weltweit in verschieden Kontinenten hintereinander die Phase 3. Die ersten Ergebnisse aus diesen Studien erwartet Telix etwa ab Q4 2019 beginnend mit Australien, dem dann die anderen Zulassungsbereiche der Reihe nach folgen. REDECTANE ist ein Wirkstoff aus der WILEX Pipeline und kann in zwei Bereichen der Diagnostik aber auch therapeutisch eingesetzt werden. Bei der Therapeutik ist der Antikörper Girentuximab etwas anders aufgebaut je nachdem welche Zielvorgaben man erfüllen möchte.
Nach der angesprochenen Optimierung des Wirkstoffes durch Telix befindet sich REDECTANE in der aktiven Phase 3 der Bilddiagnostik für die klinische Entwicklung. In der therapeutischen Anwendung befindet man sich bald in der Phase 2 und das Ende der Rekrutierung der Phase 2 wird etwa Q4 19 erwartet. Geplant sind hier zwei Studien STARLITE 1 und STARLITE 2 mit verschiedenen Wirkstoffen. Die TLX250-Therapie (177Lu-Girentuximab) hat bei Patienten mit fortgeschrittenem metastasierendem ccRCC ohne weitere Behandlungsmöglichkeiten ein progressionsfreies Überleben von etwa 10 Monaten gezeigt. TLX250 wird im Jahr 2019 in Kombination mit einer Immuntherapie mit weiteren Studien beginnen. Sollte die Entwicklung von (177Lu-Girentuximab) normal und positiv verlaufen folgt eine Phase 3 Studie und der Zulassungsprozess.
Telix rechnet hier mit Einnahmen von ca. 400-500 Millionen USD, falls es in dieser Anwendung zu einer Zulassung kommt. Erste Einnahmen dürften wir in der therapeutischen Anwendung etwa ab 2023 erwarten.
In Japan wird ein ganz neues Verfahren der Theranostic Anwendung in Zusammenarbeit mit Nihon Medi-Physics mit Beteiligung von REDECTANE erprobt. Eine Ausdehnung auf andere Gebiete ist demnach bei einem positivem Verlauf sehr wahrscheinlich. Die Umsatzbeteiligung für HP beträgt in der therapeutischen Anwendung ca. 5%. In der diagnostischen Anwendung beträgt dieser Anteil immer 30%.
Was sind Theranostics?
Theranostics sind neue Technologien die Diagnose und Therapie miteinander verbinden, dabei werden die Wirkstoffe dementsprechend konzipiert. Telix mit seinem Partner in Japan geht dabei folgenden Weg:
Zur Bekämpfung von Krebszellen möchte man Radionukliden einsetzen. Bestimmte Antikörper, die die Krebszelle erkennen, werden dann mit solch einem radioaktiven strahlenden Teil besetzt. Als Radionuklid oder radioaktives Nuklid bezeichnet man ein Nuklid (eine Atomsorte), wenn es instabil und damit radioaktiv ist. Der Antikörper wandert zur Krebszelle und die Strahlung des radioaktiven Teils zerstört dann die Krebszelle. Theranostics von Telix sind mit 225 Ac besetzt und dies ist ein Alpha-Emitter.
Im Vergleich zu anderen Arten von Radionukliden haben Alpha-Emitter eine relativ höhere Energie, um Krebszellen zu schädigen und einen kürzeren Strahlungsbereich, um Schäden an den peripheren normalen Zellen zu minimieren. Daher wird erwartet, dass 225Ac, ein alpha-emittierendes Nuklid, ein erhebliches klinisches Potenzial für die Behandlung von Krebs mittels nuklearmedizinischer Techniken aufweist. Die Parteien werden zusammenarbeiten, um die neuartige Linker-Chemie von Nihon Medi-Physics Co., Ltd. auf die Anti-CAIX-Antikörper von Telix anzuwenden und gemeinsam Proof-of-Concept-Studien durchzuführen.
In der Bilddiagnostik befindet man sich wie oben schon angesprochen nun in Phase 3 und das Ende der Rekrutierung in Europa und Australien wird etwa Q4 20 erwartet. Danach folgt die Auswertung und der Zulassungsprozess.
Die Umsatzschätzung für die diagnostische Anwendung wird mit etwa 250 Millionen USD angegeben. Neben der Anwendung bei Nierenkrebs sind nun auch neue Anwendungsgebiete verschiedener Krebsarten wie Hodenkrebs in der Diskussion bzw. in der vorklinischen Erprobung. Es besteht eine realistische Chance, dass dann zusätzlich noch mal der gleiche Betrag langfristig für die Indikationserweiterung dazu kommen. Die sehr hohe Umsatzbeteiligung für HP beträgt hier 30% vom Nettoerlös. Und man befindet sich schon in der Phase 3. Das Rekrutierungsende wird in einigen Ländern schon Ende 2019 erwartet, dem dann sukzessive die anderen Gebiete folgen werden. Erste Einnahmen bis zu ca. 75 Millionen USD dürften etwa ab 2021/22 nach der Zulassung erwartbar sein. Dies dürfte ein längerer Rollout der Gebiete werden, da die Beendigung und damit die Ergebnisse der Studien nacheinander erfolgen. Nach Australien und Japan folgt nun Europa und dann die USA. Langfristig dürfte eine Verdoppelung dieser Umsatzbeteiligungen möglich sein, da laufend weitere Anwendungsgebiete und neue Verfahren diskutiert und durchgeführt werden.
Letzte Woche kam von Telix, dass die Corona bedingten Einschränkungen nicht mehr bestehen und alle Studien wieder aktiviert worden sind.
What does this mean for Telix?
The BT designation means that the FDA will work closely with Telix to provide guidance on the development of its product TLX250-CDx. The product is being developed to determine if “indeterminate renal masses” identified in MRI or CT imaging are either clear cell renal cell cancer or non-clear cell renal cell cancer. It does this using positron emission tomography imaging, which is an imaging technique that uses radioactive substances to visualise and measure metabolic processes in the body.
According to the announcement, the new BT designation offers Telix a number of significant benefits. These include eligibility for FDA’s ‘Fast Track’ designation and more frequent and intensive interactions with the FDA. It also gives the company the opportunity to submit a ‘rolling’ biological licence application for the product, which allows it to submit its application in separate modules to streamline the approval process.
In order to achieve BT designation, Telix needed to provide preliminary clinical evidence that demonstrated its product could have substantial improvement on at least one clinically significant endpoint over available care. This means that the Telix product has shown positive results.
Jul 1, 2020
RedHill Biopharma to Present at the Sachs Novel Coronavirus Investment Forum
PDF Version
TEL-AVIV, Israel and RALEIGH, N.C., July 01, 2020 (GLOBE NEWSWIRE) -- RedHill Biopharma Ltd. (Nasdaq: RDHL) (“RedHill” or the “Company”), a specialty biopharmaceutical company, today announced that Mr. Gilead Raday, RedHill's Chief Operating Officer, will present the Company’s ongoing Phase 2/3 development programs with opaganib (Yeliva®, ABC294640)1 and RHB-107 (upamostat, WX-671)2 for COVID-19 at the Sachs Digital Novel Coronavirus Investment Forum, taking place July 7-9, 2020.
The presentation will be available on the Company's website, http://ir.redhillbio.com/events on July 7, 2020 for a period of 30 days.
In addition, Mr. Raday will participate in a panel discussion on the topic of ‘Potential Therapeutic Strategies to Prevent and Conquer Covid-19’ on Wednesday, July 8, 2020, at 10:15 a.m. EDT/4:15 p.m. CET. The live panel can be accessed by conference attendees or by registering via https://www.sachsforum.com/ncif-registration.html.
About Opaganib (ABC294640, Yeliva®)
Opaganib, a new chemical entity, is a proprietary, first-in-class, orally-administered, sphingosine kinase-2 (SK2) selective inhibitor with anticancer, anti-inflammatory and anti-viral activities, targeting multiple oncology, viral, inflammatory and gastrointestinal indications. By inhibiting SK2, opaganib impacts multiple cellular pathways which are associated with cancer growth, viral replication and pathological inflammation.
Pre-clinical data have demonstrated both anti-inflammatory and anti-viral activities of opaganib, with the potential to reduce lung inflammatory disorders, such as pneumonia, and mitigate pulmonary fibrotic damage. Several prior pre-clinical studies support the potential role of SK2 in the replication-transcription complex of positive-sense single-stranded RNA viruses, similar to coronavirus, and its inhibition may potentially inhibit viral replication. Pre-clinical in vivo studies3 have demonstrated that opaganib decreased fatality rates from influenza virus infection and ameliorated Pseudomonas aeruginosa-induced lung injury by reducing the levels of IL-6 and TNF-alpha in bronchoalveolar lavage fluids.
Opaganib was originally developed by U.S.-based Apogee Biotechnology Corp. and completed multiple successful pre-clinical studies in oncology, inflammation, GI and radioprotection models, as well as a Phase 1 clinical study in cancer patients with advanced solid tumors.
Opaganib received Orphan Drug designation from the U.S. FDA for the treatment of cholangiocarcinoma and is being evaluated in a Phase 1/2a in advanced cholangiocarcinoma and in a Phase 2 study in prostate cancer. Opaganib is also being evaluated for the treatment of coronavirus (COVID-19).
The development of opaganib has been supported by grants and contracts from U.S. federal and state government agencies awarded to Apogee Biotechnology Corp., including from the NCI, BARDA, the U.S. Department of Defense and the FDA Office of Orphan Products Development.
About RHB-107 (upamostat)
RHB-107 is a proprietary, first-in-class, orally administered potent inhibitor of several serine proteases targeting cancer, inflammatory lung diseases, and gastrointestinal diseases. RedHill is currently pursuing a Phase 2/3 clinical development program with RHB-107 for COVID-19. RHB-107 has undergone several Phase 1 studies and two Phase 2 proof-of-concept studies demonstrating its clinical safety profile in over 300 patients across 10 clinical studies, including two completed Phase 2 studies in oncology patients and several Phase 1 studies in healthy volunteers and oncology patients. These studies helped establish the safety and tolerability of RHB-107 in humans. RedHill acquired the exclusive worldwide rights to RHB-107, excluding China, Hong Kong, Taiwan, and Macao, from Germany’s Heidelberg Pharmaceuticals
https://www.edisongroup.com/publication/...velopments-coming-up/27194
https://www.focus.de/gesundheit/news/...-rekordwerte_id_11991715.html
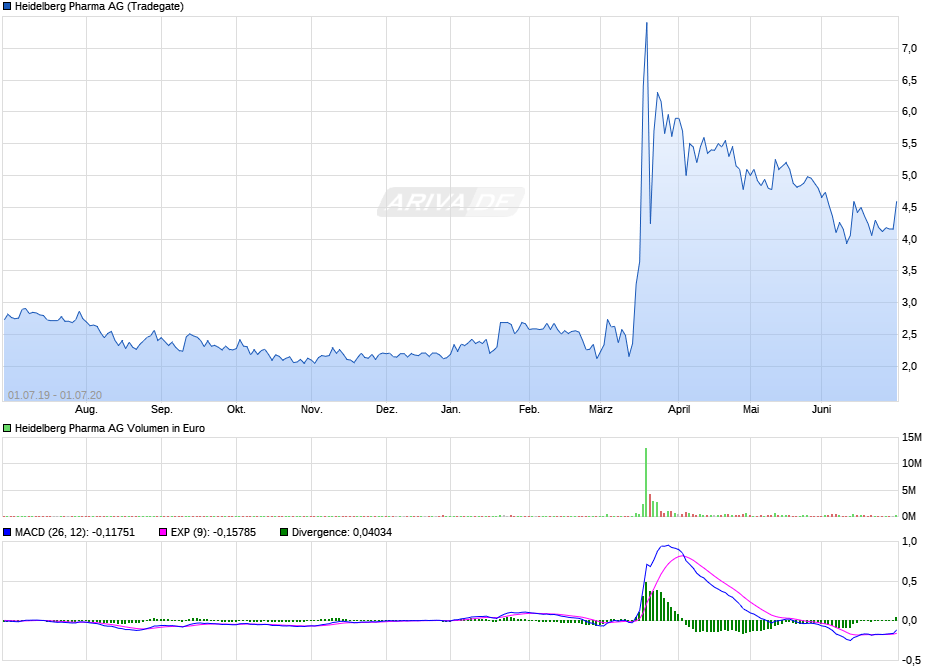
die 4 war wohl ein guter Boden zum nachkaufen, Chart schaut für mich nach einem Up-Move aus.
Vielleicht in einer wiederholten 1-5 Variante, wobei die 5 immer für große Übertreibungen bekannt ist;))
Lg
Könnte es sein, dass auch für HP Fabriken geplant sind?
morgen bei der Präsentation Omega Fund Management (US), Inc dabei, übermorgen Zahlen von HPHA, wurde das Kürzel schon für die Nasdaq geändert;) Lg
https://www.sachsforum.com/uploads/5/1/9/6/...431/ncif_agenda_web.pdf
11:00 AM
Break
16:15
Potential Therapeutic Strategies to Prevent & Conquer Covid-19 Panel
Co-Chaired by:
Rao Movva, Novartis Distinguished Scientist & Independent Pharma & Biotech Advisor Vincent Ossipow, Partner, Omega Fund Management (US), Inc.
Panelists:
Akil Jackson, Medical Director, BerGenBio ASA Christoph Esslinger, CSO, Memo Therapeutics AG Gilead Raday, COO, RedHill Biopharma Ltd.
Hella Kohlhof, CSO, Immunic Therapeutics
Josef Penninger, Co-Founder of APEIRON Biologics AG & JLP Health/ Director o
Telix Pharmaceuticals Limited ,
today announced it has entered into a strategic collaboration agreement with Hayward,
USA based RefleXion Medical, a therapeutic oncology company pioneering biology-guided
radiotherapy1 (BgRT) as a new modality for treating all stages of cancer, to investigate the clinical
utility of combining the companies’ technologies to improve treatment for high-risk or recurrent
prostate and aggressive kidney cancers.
muss mein Urlaub weiter genießen;) Lg
Auf Seite 58 vom 07.07.20
https://ir.redhillbio.com/static-files/...5f4d-4f71-9584-b7958715e8d5
RHB-107 (upamostat)
Investigational new drug
Potential first-in-class small molecule targeting oncology, inflammatory lung diseases and GI indications
Ongoing development program for COVID-19
Agreement with the National Institute of Allergy and Infectious Diseases (NIAID) to evaluate RHB-107 possible mechanism of action and potential activity against SARS-CoV-2 infection (COVID-19)