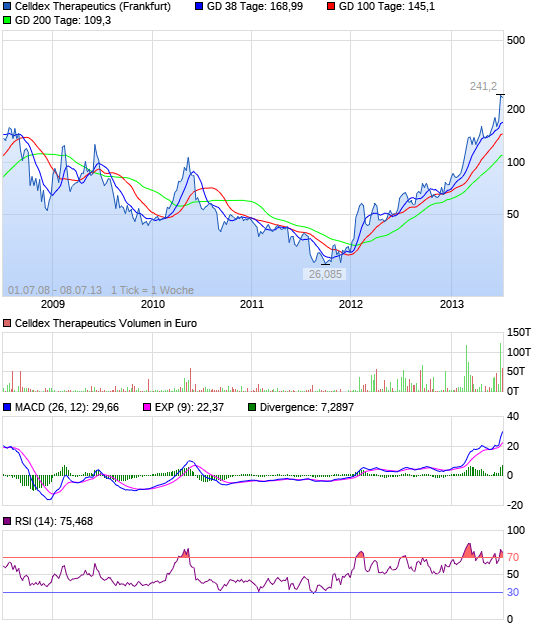
Celldex Therapeutics garantierte 100 % bis 06/10
langfristig gesehen geht das in Ordnung
halte persönlich keine Aktien mehr,
steig aber vor 2014 wieder ein
Unternehmen und günstig bewertet:
- Infos zur Bilanz: Verlust i´n Q1/2012 = 13 Mio USD + 92,4 MioUSD Cash, d.h. bestens finanziert für noch 2 Jahre
- Pipeline (nur die zwei wichtigsten):
CDX-110: Vaccine for EGFRvIII-expressing tumors = ist eine Immunotherapie die gegen EGFRvIII gerichtet ist, eine Mutation des epidermal growth factor receptor (EGFR und vailidertes Target in der Krebstherapie. EGFRvIII wird in gesundem Gewebe kaum gefunden, d.h. gesundes Gewebe sollte bei einer Theraopie kaum betroiffen sein. Zellen, die EGFRvII exprimieren, haben eine hööhere Wahrscheinlichkeit des unreguliertes Wachstums. CDX-110 aktiviert das menschluiche Immunsystem gegen Tumrozellen, die EGFRvIII exptrimieren.
"... the initial results from CDX-110 in the treatment of glioblastoma multiforme are encouraging, with an overall survival (OS) of 24.6 months compared to 15.2 months (historical cohort)."
CDX-011: "Preliminary results suggest that CDX-011 induces impressive response rates compared to current, available therapies in patients with advanced, refractory breast cancers with high GPNMB expression (expression in ≥25% of tumor cells). In this high expressing patient population, treatment with CDX-011 resulted in a 32% overall response rate (ORR; includes confirmed and unconfirmed responses), whereas treatment with Investigator's Choice (IC) single-agent chemotherapy resulted in a 13% ORR. CDX-011 also demonstrated strong response rates in patients with triple negative breast cancer across all levels of GPNMB expression (CDX-011 ORR of 21%; IC ORR of 0%), where treatment options are extremely limited. In addition, in patients with triple negative breast cancer who also highly express GPNMB, greater activity was observed (CDX-011 ORR of 36%; IC ORR of 0%). The ORR across all levels of GPNMB expression was 19% for the CDX-011 arm and 14% for the IC arm, and a direct, positive correlation was observed between increasing levels of GPNMB expression and increased CDX-011 response rates. Based on these data, the Company believes CDX-011 has significant promise as a targeted therapy for patients with breast cancer and high expression of GPNMB, and especially for those with triple negative disease."
...und das Beste ist, der Laden wird erst mit 235 Mio Euro bewertet.:.-)
CDX-1401 - Clinical Program in NY-ESO-1 Expressing Malignancies
Our second APC Targeting Technology™ product candidate, CDX-1401, is in Phase 1/2 development for the treatment of cancers known to express NY-ESO-1, including cancers of the bladder, breast, ovary, non-small cell lung cancer, myeloma, sarcoma and melanoma. This product is intended to selectively deliver the NY-ESO-1 antigen to Antigen-Presenting Cells (APCs) for generating robust immune responses against cancer cells expressing NY-ESO-1. Initial data from this ongoing study confirm safety and immunogenicity even in patients with advanced cancer who have received multiple prior therapies.
CDX-1127 - Clinical Program for Hematologic Malignancies and Solid Tumors
CDX-1127 is an agonist anti-CD27 monoclonal antibody (mAb) that is expected to activate CD27 expressing T cells in the context of T cell receptor stimulation. In addition to the immune enhancing properties of CDX-1127, the mAb may also provide direct therapeutic effects against tumors with CD27 expression. CDX-1127 is in Phase 1 development for the treatment of B cell hematologic malignancies known to express CD27 including, but not limited to, chronic lymphocytic leukemia (CLL), Burkett’s lymphoma, mantle cell lymphoma, primary lymphoma of the central nervous system and marginal zone B-cell lymphoma and solid tumors that may be more likely to be sensitive to immune therapy: metastatic melanoma, renal cell carcinoma, hormone-refractory prostate adenocarcinoma, ovarian cancer, colorectal adenocarcinoma or non-small cell lung cancer (NSCLC).
DCVax-001 – HIV Vaccine: Celldex and Rockefeller University investigators are collaborating on an effort to develop a vaccine against HIV, the virus known to cause AIDS. The Rockefeller vaccine, called DCVax-001, is an APC-targeted vaccine consisting of a fusion protein of a human monoclonal antibody with specificity for the dendritic cell receptor, DEC-205 linked to an HIV antigen. This program has been funded through a grant from the Bill & Melinda Gates Foundation. The vaccine is currently being tested in a Phase 1 trial in healthy volunteers at Rockefeller University
CDX-1135 – Renal Disease: Celldex is developing soluble Complement Receptor Type 1 (sCR1, TP10), a specific and potent inhibitor of complement activation, for use in renal disease indications. sCR1 has previously been in clinical trials and demonstrated a good safety profile and complement inhibition. sCR1 inhibits the classical, alternative and lectin pathways of complement at both early (C3 convertase) and late (C5 convertase) stages by down-regulating the activation of these cascades. sCR1 affects only activated complement and is not consumed, but can recycle in the inhibitory process. Certain rare renal diseases involve dysregulated complement and have been associated with mutations in complement proteins including Factor H and Membrane Cofactor Protein (MCP). sCR1 has similarities to Factor H and MCP in structure and function, thus providing strong support for its potential efficacy in these diseases.
CDX-1189 - Renal Disease: Celldex is developing therapeutic human antibodies to a signaling molecule known as CD89 or Fcα receptor type I (FcαRI). CD89 is expressed by some white blood cells and leukemic cell lines, and has been shown to be important in controlling inflammation and tumor growth in animal models. Celldex has proprietary, fully human antibodies to CD89 in preclinical development. Depending upon the specific antibody used, anti-CD89 antibodies can either be activating and thus stimulate immune responses, or down-regulating and anti-inflammatory.
Celldex hat ganz schön viele Aktivitäten laufen - viele im Bereich der Immunotherapie. Am interessantesten sind natürlich die weiter fortgeschrittenen Programme Phase III gegen eine besonders bösartige Form des Hirnturmos (glioblastoma) und das Phase IIb Programm (CDX-011 is a fully-human monoclonal antibody-drug conjugate (ADC) gegen Brustkrebs. In beiden Fällen gibt es bereits vielversprechende klinische Daten.
2. Celldex Therapeutics ( http://static.cdn-seekingal'495106459-Markus-Aarnio.png static.cdn-seekingalpha.com
(Click to enlarge)
Upcoming milestones
... (automatisch gekürzt) ...
www.seekingalpha.com
Zeitpunkt: 07.09.12 09:53
Aktion: Kürzung des Beitrages
Kommentar: Urheberrechtsverletzung, ggf. Link-Einfügen nutzen - Bitte nur kurz zitieren
Rufen Geplant für 7. März 2013 um 8:30 Uhr ET
http://finance.yahoo.com/news/...report-fourth-quarter-130300453.html
Needham, Massachusetts, 28. Februar 2013 (GLOBE NEWSWIRE) - Celldex Therapeutics, Inc. ( Cldx ) wird das vierte Quartal und Jahresende 2012 finanziellen Ergebnisse am Donnerstag, 7. März 2013 freigeben, bevor die US-Finanzmärkte offen. Celldex Führungskräfte wird eine Telefonkonferenz um 8:30 Uhr ET am selben Tag veranstalten bis 2012 Finanz-und Geschäftspolitik zu erörtern und ein Update auf die wichtigsten 2013 Ziele bieten.
Die Telefonkonferenz und Präsentation wird als Webcast live über das Internet und kann durch Anmeldung an der Veranstaltungskalender unter "News & Events" des Celldex Therapeutics Website zugegriffen werden www.celldextherapeutics.com . Der Anruf kann auch durch die Wahl 888-350-0137 (innerhalb der USA) oder 970-315-0478 (außerhalb der USA) abgerufen werden.
Eine Aufzeichnung der Telefonkonferenz wird etwa zwei Stunden nach der Live-Konferenz schließt bis zum 21. März 2013. Um die Aufzeichnung zuzugreifen, wählen 855-859-2056 (innerhalb der USA) oder 404-537-3406 (außerhalb der USA). Der Passcode ist 13866763. Der Webcast wird auch auf der Website des Unternehmens archiviert werden.
Über Celldex Therapeutics, Inc.
Celldex Therapeutics ist der erste Antikörper-basierte Kombination Immuntherapie Unternehmen. Celldex verfügt über eine Pipeline von Wirkstoffkandidaten in der Entwicklung für die Behandlung von Krebs und anderen schwer zu behandelnden Krankheiten auf seinen Antikörper konzentriert Precision gezielte Immuntherapie (PTI) Plattform. Die PTI Plattform ist eine komplementäre Portfolio von monoklonalen Antikörpern, Antikörper-Therapeutika und gezielte Immunmodulatoren in optimalen Kombinationen verwendet werden, um neuartige krankheitsspezifische Wirkstoffkandidaten zu erstellen. Für weitere Informationen, besuchen Sie bitte http://www.celldextherapeutics.com .
Kontakt:
Sarah Cavanaugh
Vice President of Investor Relations &
Corp Kommunikation
Celldex Therapeutics, Inc.
(781) 433-3161
scavanaugh@celldextherapeutics.com
http://www.thestreet.com/story/11862554/1/...2-financial-results.html
Zum 31. Dezember 2012 hatte Celldex rund 64.400.000 Aktien ausstehend. Als Ergebnis unserer Finanzierungen im Januar und Februar 2013 haben wir nun 80.600.000 Aktien ausstehend.
http://beta.fool.com/smartequity/2013/03/26/...ource=eogyholnk0000001
Es hat eine deutliche Rallye der Aktienkurs von Celldex (NASDAQ: Cldx ) vor kurzem. Der Bestand wird noch zeigen positive Impulse aufgrund der starken Daten über seine Arzneimittelstudien. Diese positiven Daten macht das Unternehmen zu einem attraktiven Ziel für große Pharma-Playern wie Pfizer (NYSE: PFE ) und AstraZeneca (NYSE: AZN ). Ich glaube, dass die positive Tendenz anhält und das Celldex noch ein Kauf trotz der jüngsten Kundgebungen.
Celldex Therapeutics
Celldex ist ein biopharmazeutisches in der Entwicklung und Vermarktung von neuartigen Onkologie-Therapeutika beteiligt. Das Unternehmen konzentriert sich auf die Behandlung von Krebs und anderen schwer zu Beschwerden mit seinen Antikörper Präzession gezielte Immuntherapie Plattform zu behandeln. Es hat derzeit sieben Kandidaten in verschiedenen Stadien der klinischen Entwicklung. Das Blei Kandidat ist Rindopepimut, die eine gezielte immuntherapeutischen zur Behandlung von Front-Line Glioblastom ist. Das Medikament befindet sich derzeit in zwei klinischen Studien, reagieren Studie in Phase und Act IV Registrierung Trail in Phase III. Ein weiterer wichtiger Kandidat ist CDX-011, die derzeit in der späten Phase II zur Behandlung von Brustkrebs.
Quartalsergebnisse
Der Markt erwartete Celldex Gewinn je Aktie von -0,25 $, aber das Unternehmen um 8% verfehlt berichten, die Schuld der Gewinn verpasst, höher als erwartet R & D Kosten. Royalty Zahlungen sah auch eine Erhöhung und waren verantwortlich für den höher als erwartet einen Umsatz von $ 3.650.000. Alle Gewinne verpasst führen zu einer negativen Marktstimmung, aber das Ergebnis verpasst hat keinen Einfluss auf die langfristigen Schätzungen der Aktie, da die langfristigen Wert des Unternehmens zu seiner Pipeline gebunden ist.
CDX-301
Celldex veröffentlicht positive Daten für ihren Kandidaten CDX-301 am 14. Februar. Der Markt hat positiv auf diese Ankündigung reagiert und die Aktie rund 40% seit damals geschätzt. Laut Angaben,
"Die endgültigen Ergebnisse dieser Studie sind konsistent mit früheren klinischen Erfahrungen und zeigen, dass CDX-301 ist sehr gut verträglich und wirksam zu mobilisieren hämatopoetischen Stammzell-Populationen als Monotherapie bei gesunden Probanden.
Die Rally
Die jüngste Rallye der Aktienkurs von Celldex war in erster Linie auf positive Stimmung der Anleger zu CDX-301Phase I-Daten von der Firma im letzten Monat veröffentlicht. Die Daten zeigten, dass Test-Patienten, die das Medikament nahmen eine Erhöhung der CD34 + und dendritischen Zellen sah. Das ist positiv für die Verbesserung in der Transplantationsmedizin Ergebnisse.
Im November kündigte das Unternehmen positive Phase II-Daten für rindopepimut. Die Überlebensdaten aus drei Phase-II-Studien zeigten Nutzen als eine unabhängige Kontrolle Datensätzen verglichen. Nach Unternehmensangaben Angaben,
"In der multizentrischen Phase-2-ACT III-Studie ist die mediane Überlebenszeit 24,6 Monate von der Diagnose (21,8 Monate ab Studienbeginn) und das Gesamtüberleben bei 26% nach drei Jahren. In der Phase 2 ACT II-Studie ist die mediane Überlebenszeit 24,4 Monate von der Diagnose (20,5 Monate ab Studienbeginn) und Gesamtüberleben liegt bei 23% nach drei Jahren. In der Phase 2 ACTIVATE Studie ist die mediane Überlebenszeit 24,6 Monate von der Diagnose (20,4 Monate ab Studienbeginn) und das Gesamtüberleben bei 33% nach drei Jahren. "
Das Unternehmen hat auch angekündigt, positive Daten der Phase-II Emerge Studie für ihre Brustkrebs-Kandidaten CDX-011. Das Medikament greift die GPNMB Protein, das in erster Linie verantwortlich für die Migration, Invasion und Metastasierung von Krebs ist. Die Ergebnisse, die nach Unternehmensangaben Angaben,
'Bestätigen vorläufige Ergebnisse im Mai berichtet und stellen proof of principle mit Anzeichen einer höheren Aktivität in Patienten-Untergruppen mit hoher GPNMB Ausdruck (Ausdruck in e25% der Tumorzellen), einschließlich derjenigen mit triple negativem Krankheit. "
Bottom Line
Die mittlere Sell-Side-Kursziel für Celldex ist 15,6 $, ein Aufwärtspotenzial von rund 30% auf den aktuellen Preis. Bei einer Marktkapitalisierung von rund 900 Millionen Dollar ist das Unternehmen auch ein gutes Akquisitionsziel. Es gibt einen großen Patent-Lücke im Markt, weil eine Reihe von großen Pharma-Spielern konfrontiert sind Umsatzrückgänge aufgrund auslaufender Patente. Entdeckung eines Medikaments ist ein ziemlich langer und teurer Prozess. Dies macht kleine Unternehmen mit vielversprechenden Kandidaten wie Celldex, ein attraktives Übernahmeziel.
Pfizer (NYSE: PFE ) und AstraZeneca (NYSE: AZN ) könnte Interesse an einer Small-Cap-Unternehmen wie Celldex. Pfizer steht vor Ablauf des Patentschutzes auf Lipitor und AstraZeneca steht vor der Konkurrenz durch Generika zu seinem Stern Medikament Seroquel IR. Der neue CEO von AstraZeneca, Pascal Soriot wird versuchen, um den fallenden Umsatz von AstraZeneca durch die Konzentration auf neue Wirkstoffkandidaten zu machen.
Die schwache Pipeline des Unternehmens erhöht die Wahrscheinlichkeit, dass das Unternehmen Interesse am Erwerb einer kleinen Firma wie Celldex mit einer Marktkapitalisierung unter $ 1 Milliarde. In einer kürzlich durchgeführten strategischen Wandel hat das Unternehmen auch beschlossen, sich auf drei Schwerpunkte: Herz-Kreislauf-, Krebs-und Stoffwechselerkrankungen. Pfizer hat bereits eine ziemlich starke Onkologie-Pipeline mit führenden Kandidaten PD-0332991 in Phase-II-Studien zur Behandlung von Brustkrebs. Nach Sell-Side-Schätzungen kann das Medikament habe ein Umsatzpotenzial von rund 5 Milliarden Dollar.
http://seekingalpha.com/article/...y-affect-share-prices?source=yahoo
Für eine Entwicklungsphase Biotechnologie, bleiben flott ist nie eine gegeben, und die Optionen sind nicht schön, wie man das macht. Jedes Unternehmen hat seine eigene Strategie und jede Strategie hat ihre eigenen Vor-und Nachteile. In diesem Beitrag werde ich über vier vielversprechende Biotechnologieunternehmen, die kämpfen, um ihre Medikamente auf den Markt zu bekommen sind, die jeweils ein führender einen anderen Ansatz, wie man ohne wesentliche Einnahmequelle überleben gehen. Ich werde darauf hinweisen, die Vorteile und Nachteile der einzelnen Ansätze, um Ihnen eine Idee geben, wie jeder könnte der gegebenen Aktienkurs des Unternehmens im Falle von Erfolg oder Misserfolg beeinflussen.
Celldex ( Cldx ) Strategie: Halten Sie einen schönen Hauptstadt Kissen
Bei $ 900M ist Celldex recht groß für ein Unternehmen ohne Umsatz. Das Handelsvolumen ist somit viel höher als die meisten seiner Konkurrenten, wie Liquidität in den Aktienkurs. Celldex hat sechs klinischen Studien, die derzeit durchgeführt, die wichtigste von denen der Phase-III-Studie für seine interessanterweise genannten brain Krebsmedikament Rindopepimut für Glioblastom ist. Die Studie mit dem Codenamen ACT IV wird 374 Krebspatienten Ende der Einschreibung sind, kostete das Unternehmen einen geschätzten $ 60M und Celldex Schätzungen Einschreibung bis Ende dieses Jahres abgeschlossen werden, wobei die Ergebnisse kommen in bis zu 24 Monate später.
Die Firma versucht, die Big Pharma Ansatz nehmen und Pfizer ( PFE ) zu finanzieren ACT IV und ein Abkommen zu tun, wurde im April 2008 unterzeichnet, aber es fiel durch im November 2010 und alle Rechte an Rindopepimut wurden Celldex zurückgegeben (siehe Seite 2 auf den Link oben), die jetzt die Finanzierung wird die Studie auf seine eigenen. Celldex auch versucht, Fast-Track in der Phase II und kombinieren Sie es in Phase III, Zeit und Geld durch Strukturierung der Studie mit einer Kontrollgruppe, also Gehirn Krebspatienten, die nicht bekommen Rindopepimut würde und vergleichen Sie die beiden Gruppen zu speichern. Im Dezember 2008 scheiterte dieser Versuch und Celldex war gezwungen, den Versuch strikt einer Phase II zu ändern, da die Patienten randomisiert der Kontrollgruppe einfach fallengelassen aus der Studie, aus verständlichen Gründen. Damit Verknüpfung nicht funktionieren.
Celldex Strategie seither zu pad seine Ressourcen und die Bilanz mit viel Bargeld durch präventive Eigenkapitalfinanzierungen gewesen. Seit Anfang 2013 hat es über $ 114M in zwei garantierten öffentlichen Angebot im Januar und Februar mit über 16 Millionen Aktien erhöht. Die Begeisterung für die Produktpalette des Unternehmens Kandidaten, vor allem Rindopepimut hat Cldx Aktien im Gesicht der beiden Verdünnungen angehoben, um sie unmerklich. In der Tat, seit Januar hat die Aktie 67% sprunghaft angestiegen. Die Frage ist, warum? Dafür haben wir einen Blick auf Rindopepimut nehmen.
Celldex Flaggschiff-Pipeline Medikament ist eine Immuntherapie, die ein Protein namens EGFRvIII, in etwa zielt ein Viertel der 15.000 jährlichen Glioblastom Fälle in den USA. Phase-II-Studie Daten zeigten (Seite 18) das Gesamtüberleben bei 24,6 Monaten über 15,2 Monate in der Kontrollgruppe, und die progressionsfreie Überlebenszeit von 8,5 Monaten ab Diagnose. Wenn diese Daten in Phase III repliziert werden, ist die FDA-Zulassung wahrscheinlich, und die Anleger wissen. Auch einen Beitrag zur Optimismus sind Celldex geschätzten Zeitrahmen für die Phase III bei der Einschreibung voraussichtlich in weniger als einem Jahr, die einen Großteil der $ 60M Kosten bedeutet haben wird bis dahin werden abgedeckt wurden. Drittens, ironisch, ist wahrscheinlich die Tatsache, dass Pfizer ausgestiegen seiner Lizenzvereinbarung, wodurch alle Rechte in Celldex Hände Rindopepimut verwandt. Dies bedeutet, dass sie alle die Einnahmen aus dem Verkauf zu sehen, ob und wann das Medikament zugelassen ist. Dies, kombiniert mit der Tatsache, dass die Phase III voran, ist das, was kann treibt Cldx in letzter Zeit.
Die Vorteile für die präventive Marktkapitalisierung Ansatzes sind, dass ihre Finanzen gesund aussehen und seiner Bilanz fest. In der Tat, mit $ 117M in bar und weniger als $ 12M in Gesamtverschuldung (1,3% der Marktkapitalisierung), sieht Cldx gesund und attraktiv für Investoren.
Die Nachteile sind jedoch, dass der gute Eindruck nur, dass, und alles hängt von der Phase III ist. Ab sofort wird das Lager für den Erfolg preiswert, aber Zulassung ist ca. 3 Jahre weg, geschätzt auf November 2016 . Erwarten Sie ein Zusammenbruch der letzten Parabel und kaufen, wenn und wann es $ 8 bricht.
Celldex Therapeutics (CLDX: Nasdaq) Nach Brean Kapital ($ 15,53, 11. Juni 2013) Wir erhöhen unser Kursziel auf Celldex Therapeutics bis $ 19 aus $ 14 aufgrund seiner starken klinischen Daten zu Datum und robuste Pipeline. Wir bewerten Celldex (Ticker: CLDX) ...
http://ir.celldextherapeutics.com/releasedetail.cfm?ReleaseID=774822
Von James Giaquinto 07.05.13 - 14.00 Uhr EDT
inShare
http://www.thestreet.com/story/11970414/5/...-traded-by-insiders.html
Celldex Therapeutics (CLDX) - FREE Research Report
Catlin Avery W, die Sr. VP & CFO bei Celldex THERAPT ist gekauft 500 Aktien bei $ 5,95 pro Aktie für einen Gesamtwert von $ 2,975.00 am 1. Juli 2013 beginnen. Nach dieser Transaktion im Besitz der Sr. VP & CFO 14.109 Aktien bedeutet, dass der Anteil von 3,67% wurde mit der 500-Anteilserwerb erhöht.
Marucci Anthony S, der President & CEO bei Celldex THERAPT ist gekauft 500 Aktien bei $ 5,95 pro Aktie für einen Gesamtwert von $ 2,975.00 am 1. Juli 2013 beginnen. Nach dieser Transaktion im Besitz der President & CEO 43368 Aktien bedeutet, dass der Anteil von 1,17% wurde mit der 500-Anteilserwerb erhöht.
Die Aktien zuletzt bei 20,74 $ gehandelt, bis 2,09 $ oder 11,21%, da die Insider-Transaktionen.
Das durchschnittliche Volumen für Celldex Therapeutics hat 1.600.000 Aktien pro Tag in den letzten 30 Tagen. Celldex Therapeutics hat eine Marktkapitalisierung von $ 1,5 Milliarden Euro und ist Teil des Gesundheitswesen und Pharmaindustrie. Anteile sind bis 177.94% seit Jahresbeginn per Ende des Handels am Mittwoch dem Laufenden.
Celldex Therapeutics, Inc., ein biopharmazeutisches Unternehmen, fokussiert sich auf die Entwicklung, Herstellung und Vermarktung neuartiger Therapeutika für das Gesundheitswesen vor allem in den Vereinigten Staaten. Derzeit gibt es 7 Analysten, dass Rate Celldex Therapeutics ein Kauf, keine Analysten bewerten es ein Verkaufssignal, und keiner rate it a hold.
Exklusives Angebot: Holen Sie sich die neueste Aktienanalyse auf CLDX - FREE
TheStreet Quant Bewertungen Preise Celldex Therapeutics als verkaufen. Das Unternehmen Schwächen können in mehreren Bereichen, wie seine schwachen operativen Cash-Flow und der sich verschlechternden Nettoeinkommen gesehen werden. Holen Sie sich die volle Celldex Therapeutics Ratings Bericht TheStreet Quant Bewertungen jetzt.
Aktien zu kaufen: TheStreet Quant Ratings hat eine Handvoll Aktien, die möglicherweise in den nächsten 12 Monate verdreifachen kann identifiziert. Erfahren Sie mehr.
http://finance.yahoo.com/news/...nces-issuance-seminal-110000546.html
Patent Bietet Broad-Based geistiges Eigentum Plattform für CDX-1127
Phillipsburg, NJ, 10. Juli 2013 (GLOBE NEWSWIRE) - Celldex Therapeutics, Inc. ( CLDX ) gab heute bekannt, dass das United States Patent and Trademark Office (USPTO) erteilt hat US-Patent Nr.: 8.481.029 mit dem Titel "Human Immuntherapien unter Verwendung eines CD27 Agonist allein oder in Kombination mit anderen Immunmodulatoren ", die im Großen und Ganzen unterstützt der Gesellschaft Produkt-Kandidat CDX-1127. CD27, ein Signalmolekül auf T-Lymphozyten exprimiert, kann wirksam mit aktivierenden Antikörpern gegen potente anti-Tumor-Reaktionen und aufgrund der eingeschränkten Expression und Regulation von CD27 induzieren manipuliert werden, kann in weniger Toxizität-Überwindung eine zentrale Barriere führen, dass andere Ziele in diese Klasse konfrontiert. CDX-1127 ist ein vollständig humaner monoklonaler Antikörper (mAb), die CD27 richtet und befindet sich derzeit in Phase 1 der klinischen Entwicklung zur Behandlung von soliden Tumoren und malignen hämatologischen Erkrankungen.
Das Patent mit der University of Southampton zugeordnet und Celldex hat eine exklusive Lizenz für dieses Patent auf die Ausführung eines exklusiven Lizenzvertrag mit der University of Southampton die menschliche Antikörper gegen CD27 entwickeln im November 2008. Das Patent umfasst 18 Claims verschiedene Methoden der Behandlung von Krebs mit Hilfe agonistischen Anti-Human-CD27-Antikörper und bezieht sich unter anderem direkt Celldex die CD27 Antikörper Programm und therapeutische Verwendung von Celldex Antikörper-CDX-1127.
"Wir sind weiterhin gute Fortschritte machen fortschreitenden CDX-1127 und Sicherung dieses Kernstück des geistigen Eigentums ist eine wichtige Errungenschaft, da wir unser klinisches Programm in soliden Tumoren und vollständige Dosis-Eskalations-Studien in malignen hämatologischen Erkrankungen erweitern", sagte Tibor Keler, Senior Vice President und Chief Scientific Officer von Celldex Therapeutics. "Dieses Patent auch direkt spricht für die Bedeutung der innovativen Arbeit von Professor Martin Glennie und sein Team an der Universität von Southampton in Targeting Mitglieder der TNF-Rezeptor-Superfamilie-die wir glauben, ist in schnellen Übersetzung von spannenden immunstimulierende Antikörper in der Klinik was. "
Auf das Jahr 2013 American Association of Cancer Research Annual Meeting, in vitro menschliche Daten wurden vorgestellt, gezeigt, dass CDX-1127 potente Aktivierung von T-Zellen durch die Induktion ihre Verbreitung und Veröffentlichung von wichtigen immunmodulierende Zytokine auslöst. Am wichtigsten ist, zeigten die Daten, dass diese Aktivierung wird dringend geregelt, welche begrenzt Sicherheitsbedenken im Zusammenhang mit nicht-spezifische Stimulation des Immunsystems, dass ähnliche Kandidaten in dieser Klasse konfrontiert. Diese Erkenntnis haben sich weiter durch das günstige Sicherheitsprofil gesehen in unserer laufenden Multi-Dosis Phase 1 der klinischen Studie bisher unterstützt werden. Im festen Tumor Arm hat Dosissteigerung abgeschlossen und CDX-1127 wurde bestimmt, dass gut verträglich, auch auf der höchsten Dosis. Das Unternehmen hat kürzlich initiierte die Expansion Teil der Studie mit Kohorten in metastasiertem Melanom und Nierenzellkarzinom. Die hämatologischen Tumor Arm weiterhin Patienten in der Dosis-Eskalations-Teil der Studie einschreiben und rechnet damit, in Expansion Kohorten bis Ende des Jahres zu verschieben.
Über Celldex Therapeutics, Inc.:
Celldex entwickelt zielgerichtete Therapeutika zu verheerenden Krankheiten, für die verfügbaren Behandlungen unzureichend ansprechen. Unsere Pipeline ist aus einem firmeneigenen Portfolio von Antikörpern und Immunmodulatoren allein und in strategische Kombinationen, neue, krankheits-spezifische Therapien, induzieren verstärken oder unterdrücken die körpereigene Immunantwort zu erstellen verwendet gebaut. Besuchen http://www.celldextherapeutics.com .
Kontakt:
Kontakt zum Unternehmen:
Sarah Cavanaugh
Vice President der IR & Corp Comm
Celldex Therapeutics, Inc.
(781) 433-3161
scavanaugh@celldextherapeutics.com